- Blog
- Neat video 4 crack
- Fm tilt stereo tools
- Jimmy swaggart ministry
- Encore enuwi belkin n150 micro wireless usb adapter
- Jm digital works
- Swedish remington rolling block rifle bayonet
- Tame impala let it happen ending
- Moliken population quantumwise
- Transferring to preferred server ffxiv
- Most accurate sega saturn emulator
- Cool edit pro 2-0 free download
- Worldworksgames for malifaux
These studies suggest thatthrough the application of defect, it is possible to alter theelectronic properties of graphene that leads to changes in theirchemical reactivity. due tothe fact that imperfection at the edges provide more danglingbond for molecules attachment. Considerableroles played by the graphene edges in improving gas sensingproperties has also been explored by Tan et al. This is dueto the present of long and reactive edges. As such, graphene has beentreated by many ways in order to enhance the reactivity withadsorbates allowing for control over the subsequent properties.GNR are more attainable to doping, chemical modificationand sensitive to structural defects and impurities. Toachieve a high figure of merit for such application requiresa materials with high reactivity. There is extensive literature on the investigation of graphenein detecting various gas and bioactive molecules. Furthermore, graphenehave a low degree of crystal defects to withstand thermalswitching. The tunable bandgap and excellentelectrical conductivity in graphene is beneficial for conducting Graphene havebeen shown to posses large surface to volume ratio and highcarrier density leading to maximize the interaction betweenits surface and adsorbates. Researchersstudying graphene since it was isolated in 2004 recognizedit potential application in detecting individuals gas molecules since it properties is similar to CNT. It is reported thatusing CNT can detect small concentration of NH3 and O2at room temperature with high sensitivity. Therefore sensor to detect possibleleakage for early detection is important. Thisgas with higher concentration are found in chemical industryand medical application. For exampleNH3 is a toxic gas which present in atmosphere naturally. Since GNR and graphene haveflat structure and larger possible surface area applying as gassensor is more appropriate than other carbon based materials.Gas molecules detection has a great significant in many areassuch as environmental and industrial inspection. As an alternative, considerable interest has gen-erated in carbon based materials like graphene to help boostsensing device performance. Workers studying gas sensor recognized the limita-tion such as low binding energy, low sensitivity and poorselectivity. Sensing exploit semiconductor materials and become thefundamental building block in the design of sensitive gassensor. Index Termsgraphene nanoribbon, adsorption, gasmolecules. We show that thedefective AGNR are sensitive to the adsorption of molecular NH3.
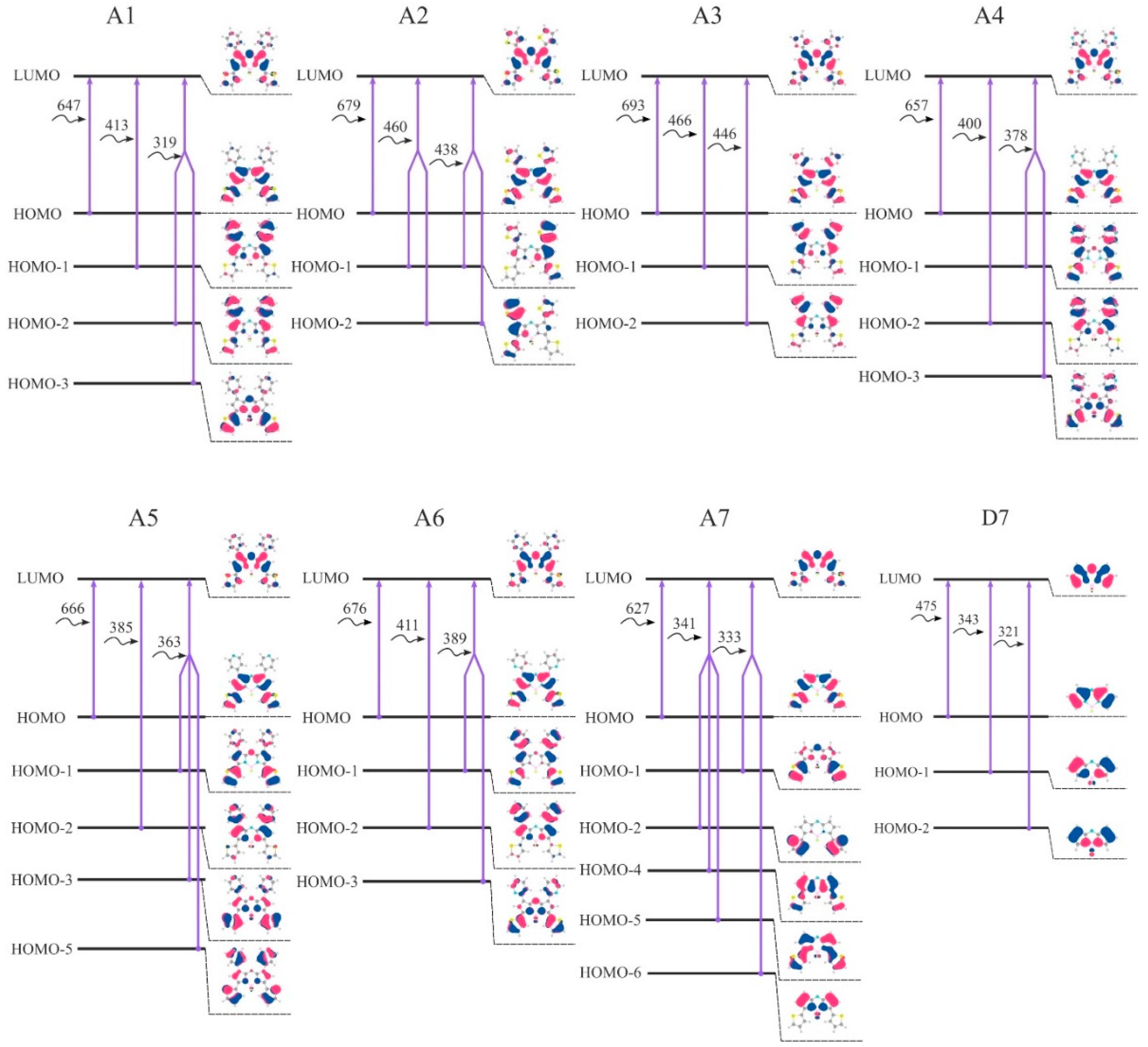
The calculatedcharge transfer shows that molecular NH3 acts as a donor whileatomic NH3, O2 and N2 acts as acceptor. Other gases havelittle effect on modifying the electronic structure. MolecularO2, N2 and NH3 adsorption at near position achieved highestadsorption energy compared to other positions. Through simulation, itis demonstrated the gas molecules adsorption energy greatlyinfluenced by its position towards the SV defects. Bothmolecular and atomic gas molecules configuration are consideredin determining the adsorption energy. The main focus is to investigate using self-consistent Extended-Huckel the adsorption geometry of the gasmolecules located at on, near and far from the SV defect. Ezaila AliasDepartment of Computer & Electronic Engineeringįaculty of Electrical EngineeringUniversiti Teknologi MalaysiaĨ1310 UTM Johor Bahru, Johor, paper reported on the structural and elec-tronics properties of armchair graphene nanoribbon (AGNR)distorted with single vacancy (SV) defect when adsorbing var-ious gas molecules including oxygen (O2), nitrogen (N2) andammonia (NH3). Zuriana Auzar, Zaharah Johari, Sakina S.H and N. Computational Study of Gas Molecules Adsorption on Defective Graphene
- Blog
- Neat video 4 crack
- Fm tilt stereo tools
- Jimmy swaggart ministry
- Encore enuwi belkin n150 micro wireless usb adapter
- Jm digital works
- Swedish remington rolling block rifle bayonet
- Tame impala let it happen ending
- Moliken population quantumwise
- Transferring to preferred server ffxiv
- Most accurate sega saturn emulator
- Cool edit pro 2-0 free download
- Worldworksgames for malifaux
